In the past decade C. elegans has become a popular model in investigating molecular effects of phytochemicals. Among these, flavonoids represent a major group of plant secondary metabolites shown to alter lifespan and stress resistance in C. elegans ( Kampkoetter et al., 2008). However, bioavailability of these compounds is rather low. Detection in worms usually requires analytical techniques such as HPLC and rather large amounts of worm material but gives no information on where in worms the compound accumulates (Kampkoetter et al., 2008). Here we report a method to detect and semi-quantify flavonoids in C. elegans by analysis of the fluorescence after derivatisation in vivo. For this purpose, we used 2-aminoethyl diphenyl borate (Naturstoff reagent A (NSRA)), which is employed in thin layer chromatography, plant histology and cell culture to enhance auto-fluorescence of plant polyphenols (Neu, 1956; Ernst et al., 2010). NSRA forms a chelate with the polyphenolic compounds, leading to a shift of the spectral band to a longer wavelength (bathochromic shift) and to an intensified signal.
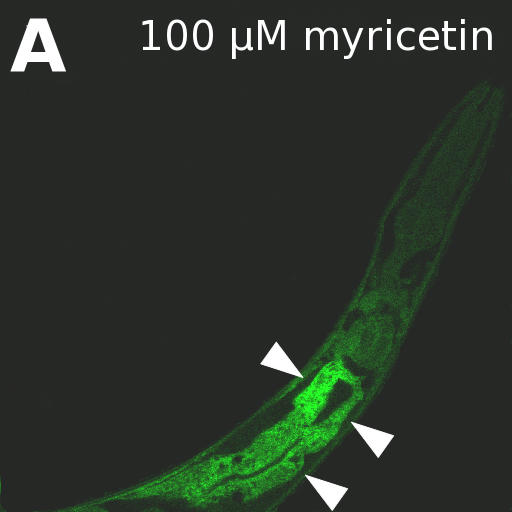
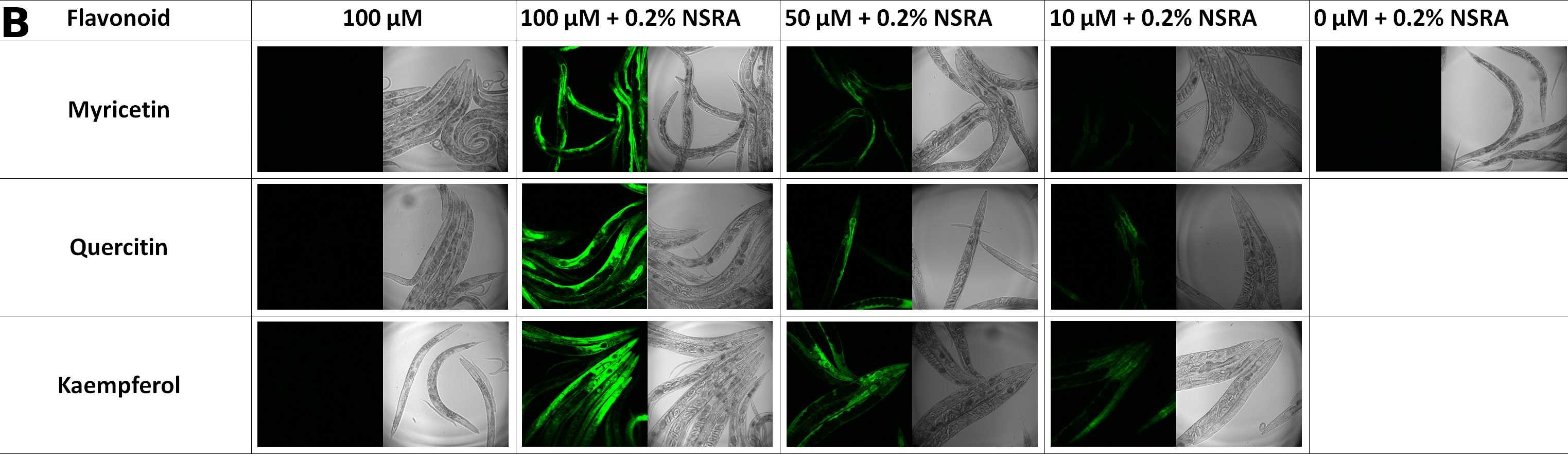
We exposed wild type N2 L4-larvae on NGM plates to the flavonoids myricetin, quercitin or kaempferol in increasing concentrations (0, 10, 50 or 100 µM) for 48 hours. Thereafter, the worms were incubated in M9 buffer containing 10% heat-killed OP50 and 0.2% NSRA for 2 hours. Subsequently, fluorescence was monitored using a confocal laser scanning microscope with excitation at 488 nm and emission at 590-620 nm (myricetin and quercitin) and at 540-560 nm (kaempferol).
NSRA selectively increased the fluorescence of the flavonoids, making them detectable in C. elegans. Highest fluorescence was always obtained in gut epithelial cells (A) as the sites of flavonoid uptake and intensity reflected the flavonoid concentrations in a dose-dependent manner (B). This shows that NSRA is an easy-to-use and cheap tool to visualize and semi-quantify flavonoids in vivo in C. elegans.
Figures
References
Ernst IM, Wagner AE, Lipinski S, Skrbek S, Ruefer CE, Desel C, Rimbach G. (2010). Cellular uptake, stability, visualization by 'Naturstoff reagent A', and multidrug resistance protein 1 gene-regulatory activity of cyanidin in human keratinocytes. Pharmacol. Res. 61, 253-258. 
Kampkötter A, Timpel C, Zurawski RF, Ruhl S, Chovolou Y, Proksch P, Wätjen W. (2008). Increase of stress resistance and lifespan of Caenorhabditis elegans by quercetin. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 149, 314-323. 
Neu R. (1957). Chelate von Diarylborsäuren mit aliphatischen Oxylalkaminen als Reagenz für den Nachweis on Oxyphenyl-benzo-gamma-pyronen. Naturwissenschaften 44, 181–182.