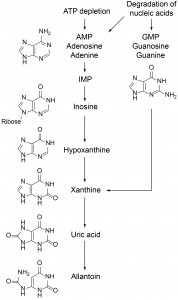
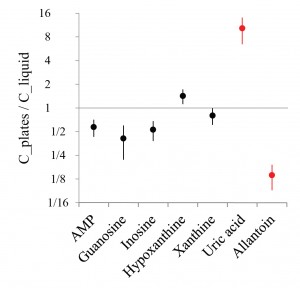
It is well documented that parental experience may profoundly influence subsequent generations. But the mechanisms of such effects have only recently started to be revealed. During the first hours of L1 starvation C. elegans release products of purine metabolism (probably resulting from ATP depletion and/or RNA degradation, Fig. 1). We discovered that the end product of the purine degradation pathway depends on the history of parents of arrested L1 larvae. When comparing exo-metaboloms of arrested L1s (L1 medium) whose parents were grown on bacterial plates vs. liquid bacterial culture we found that progeny of worms grown in liquid produce substantially more allantoin and less uric acid than progeny of worms from plates (Fig. 2). Levels of purine metabolites upstream of uric acid and many other metabolites did not show significant differences (Fig. 2). The small amount of uric acid in eggs could not account for its concentration in L1 medium, suggesting that uric acid is produced by L1 worms and not supplied in the egg. Furthermore, levels of uric acid and upstream purine metabolites were similar in eggs from worms grown on plates and in liquid culture. We conclude that in arrested L1 larvae from plate-grown mothers conversion of uric acid to allantoin is retarded and thus uric acid accumulates, while in L1 worms from liquid culture-grown mothers purine metabolism proceeds to allantoin.
Like humans, C. elegans lacks uricase, an enzyme responsible for oxidation of uric acid to allantoin (Ramazzina et al., 2006). Thus one would expect uric acid to be the final product of the purine degradation pathway. The presence of allantoin can be rationalized in two ways. First, uric acid may be oxidized to allantoin by some other enzyme, e.g., a cytochrome P450. In humans, myeloperoxidase has been shown to catalyze this step (Meotti et al., 2011). Second, uric acid is a known antioxidant (Ames et al., 1981), and its oxidation can proceed nonenzymatically, e.g., in the presence of ROS, with allantoin being one of the oxidation products.
We profiled mRNA levels in arrested L1 larvae from plate- and liquid culture-grown mothers. We found 145 genes that were different between these two groups by a factor of 3 or more (http://elegans.som.vcu.edu/~alex/purine_metabolism). Half of them were genes encoding proteins with unknown function or homology. sod-3 and sod-5 were overexpressed in L1 progeny from plates compared to liquid culture (3 and 4.1 fold difference). This might indicate that progeny from plates have better ROS protection, while liquid culture progeny are more susceptible to oxidative stress and uric acid acts there as an endogenous antioxidant, resulting in its nonenzymatic oxidation to allantoin. We then compared uric acid and allantoin levels in L1 medium of sod-3(tm760), sod-5(tm1146) and N2 whose parents were grown on plates and found no significant difference. We also measured uric acid and allantoin concentrations in L1 medium (both from plates and liquid culture) in the presence of 200 µM epicatechin, an antioxidant that was shown to reduce oxidative stress in adult C. elegans (González-Manzano et al., 2012). Epicatechin had no effect on uric acid and allantoin levels. Overall, these experiments suggest that different levels of oxidative stress may not explain differences in the uric acid/allantoin ratio.
The microarray data showed that the P450 family gene cyp-13A5 is upregulated 5.4 fold in liquid culture progeny, so we hypothesized that cyp-13A5 might catalyze oxidation of uric acid to allantoin. RNAi knockdown of cyp-13A5 in liquid culture-grown mothers did not cause a change in uric acid and allantoin levels in L1 medium of the progeny. In the same conditions unc-22 RNAi caused a partial uncoordinated phenotype, but we did not verify cyp-13A5 knockdown level by qPCR, so this result may not be conclusive.
In conclusion, we found that in arrested L1 worms whose parents were grown on plates purine metabolism stops at uric acid, while in arrested L1s whose parents were grown in liquid culture it proceeds one step further and uric acid is converted to allantoin. At present, we do not have a satisfactory explanation for this effect. Finally, we found that there was no significant difference in L1 starvation survival between these two groups of worms.
We thank Maciej Kukula for the help with HPLC-MS.
Figures
References
Ames BN, Cathcart R, Schwiers E, Hochstein P. (1981) Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: a hypothesis. Proc Natl Acad Sci U S A 78, 6858-6862. 
González-Manzano S, González-Paramás AM, Delgado L, Patianna S, Surco-Laos F, Dueñas M, Santos-Buelga C. (2012) Oxidative status of stressed Caenorhabditis elegans treated with epicatechin. J Agric Food Chem 60, 8911-8916. 
Johnson RJ, Sautin YY, Oliver WJ, Roncal C, Mu W, Sanchez-Lozada LG, Rodriguez-Iturbe B, Nakagawa T, Benner SA. (2009) Lessons from comparative physiology: could uric acid represent a physiologic alarm signal gone awry in western society? J Comp Physiol B 179, 67–76. 
Meotti FC, Jameson GNL, Turner R, Harwood DT, Stockwell S, Rees MD, Thomas SR, Kettle AJ. (2011) Urate as a physiological substrate for myeloperoxidase: implications for hyperuricemia and inflammation. J Biol Chem 286, 12901-12911. 
Ramazzina I, Folli C, Secchi A, Berni1 R, Percudani R. (2006) Completing the uric acid degradation pathway through phylogenetic comparison of whole genomes. Nat Chem Biol 2, 144-148.